Definition Of Gene Therapy Ema

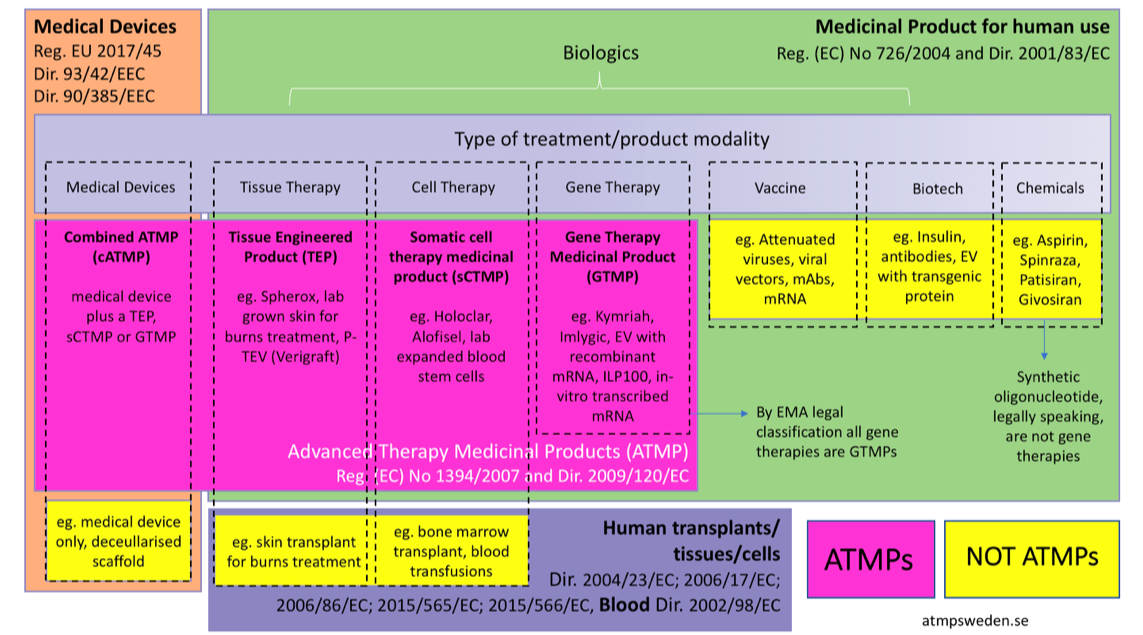
Advanced therapy medicinal products atmps are medicines for human use that are based on genes tissues or cells.
Definition of gene therapy ema. The first attempt at modifying human dna was performed in 1980 by martin cline but the first successful nuclear gene transfer in humans approved by the national institutes of health was performed in. Glybera is a recombinant adeno associated virus aav vector designed for gene therapy of lipoprotein lipase deficiency 1 while strimvelis is a genetically modified hematopoietic stem cell. The legislative framework for market authorization of human gene therapy products and other medicinal. The goal of gene therapy is to cure a genetic.
We define advanced cell therapy as all therapies in which cells are more than minimally manipulated and or their action is not homologous. Medicinal product for human use that is a gene therapy medicinal. When a gene is damaged the blueprint for the construction of the protein it codes for is imperfect. The marketing authorization of glybera and strimvelis by the european medicines agency ema marked the end of the long and often troubled road of gene therapy from biological concept to medical practice.
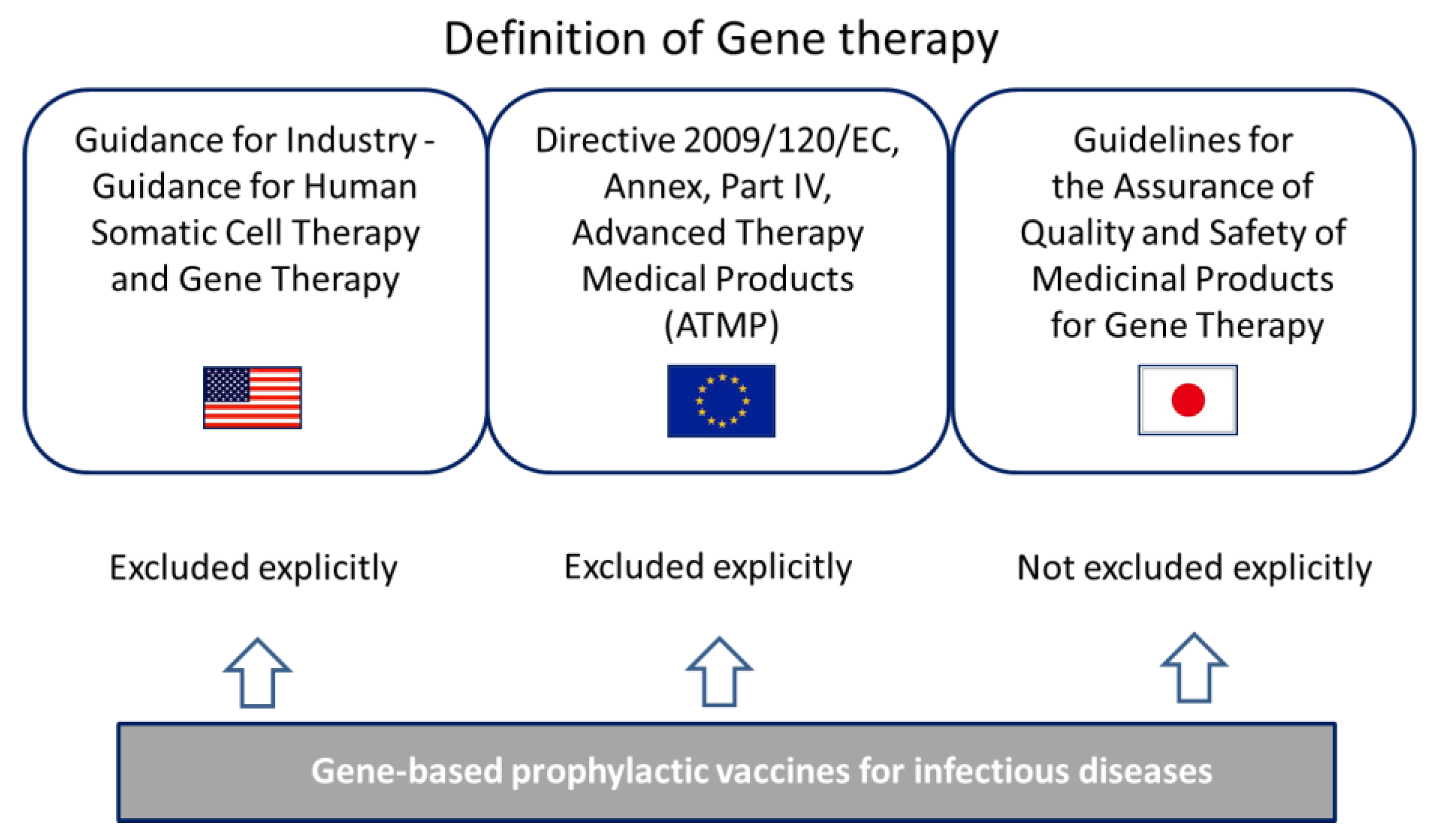
The european medicines agency s scientific guidelines on gene therapy help medicine developers prepare marketing authorisation applications for human medicines. Questions and answers on comparability considerations for advanced therapy medicinal products atmp ema cat 499821 2019 the overarching guideline for human gene therapy medicinal products is the guideline on the quality non clinical and clinical aspects of gene therapy medicinal products ema cat 80183 2014. The resulting protein is therefore abnormal. With a gene therapy vector prior to administration to the human subject as they are covered in the guideline on quality non clinical and clinical aspects of medicinal products containing genetically modified a cat gtwp 671639 2008cells em.
These contain genes that lead to a therapeutic prophylactic or diagnostic. Our definition is similar to the european medicines agency ema definition of advanced therapy medicinal product atmp. They offer groundbreaking new opportunities for the treatment of disease and injury. On the interpretation of key concepts of the definition of gene therapy medicinal product somatic cell therapy medicinal product tissue engineered product and combined advan ced therapy medicinal product.
The guidance reflects the experience gaine d in the application of the classification procedure. The final stage in the development of a gene therapy product after conducting clinical trials is the market application. The european medicines agency ema formerly known as emea is a european agency for the evaluation of medicinal products including gene therapy medicinal products. Gene therapy also called human gene transfer is a medical field which focuses on the utilization of the therapeutic delivery of nucleic acids into a patient s cells as a drug to treat disease.
Definition of advanced cell therapy trials.