Definition Of Data Integrity In Pharmaceutical Industry

It provides foundational assurance that the data the company uses to demonstrate that its products are safe and effective for their intended use are in compliance with regulatory requirements.
Definition of data integrity in pharmaceutical industry. Data integrity is the principle of generating maintaining and assuring the consistent accuracy and completeness of data in a company or manufacturing plant through the entire life cycle complying with the regulations set within the pharmaceutical industry by the food and drug administration fda. Sop on data integrity in pharmaceutical industry date. Data integrity is the degree to which data are complete consistent accurate trustworthy reliable and that these characteristics of the data are maintained throughout the data life cycle. 4 7 handwritten entries should be made in a clear legible indelible way.
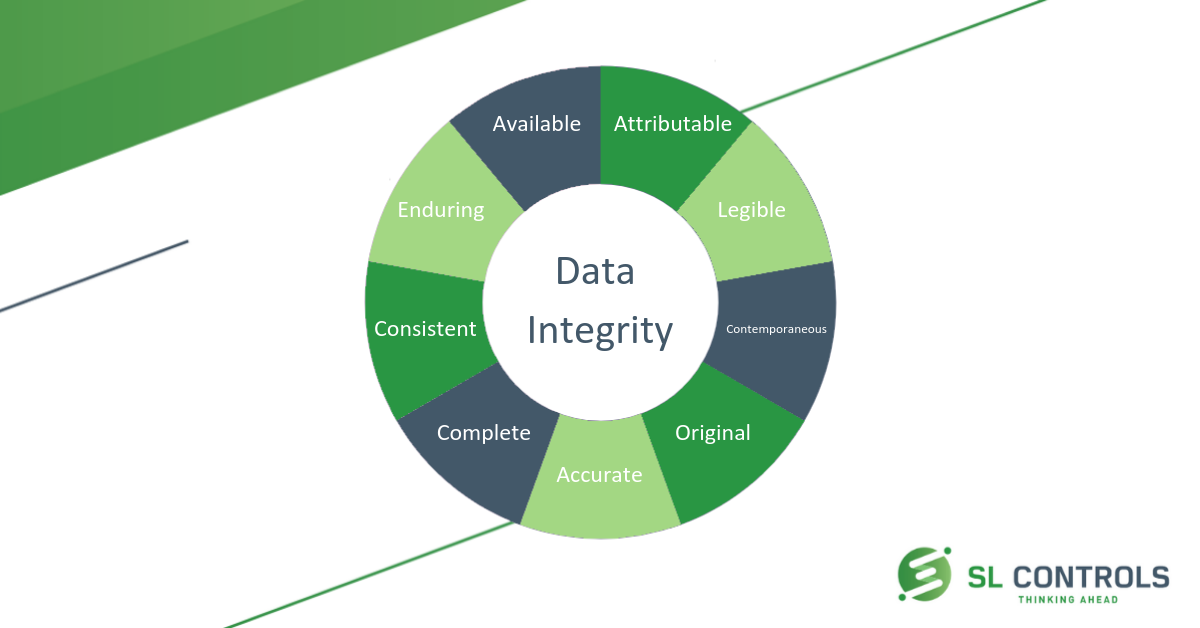
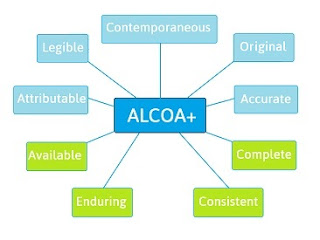
Alcoa is the data integrity standard of regulatory bodies hence the needed industry standard. Alcoa to alcoa plus for data integrity alcoa was an tool to implement the data integrity in pharmaceutical manufacturing facility but acloa made this tool more powerful and sharp. Data integrity is defined as the degree to which all data electronic paper based or hybrid are complete consistent and accurate throughout the lifecycle of the data. To lay down a procedure for data integrity as part of quality management system.
Cbe 012 v03 definition of raw data in our industry. Data integrity is the extent to which all data are complete consistent and accurate throughout the data lifecycle. Pharmastate blog 1 comment facebook twitter linkedin whatsapp objective. In case of sun pharma the stock moved up.
A data integrity program is a significant component of a company s quality system. Usfda suggests that data integrity refers to the completeness. This makes it interesting to understand data integrity from the eyes of usfda. Fda cfr 58 glps raw data means any laboratory worksheets records memoranda notes or exact copies thereof that are the result of original observations and activities of a nonclinical.
The data should be collected and maintained in a secure manner so that they are attributable legible contemporaneously recorded original or a true copy and accurate.